AI-driven pharmacometrics — PopPK, PBPK, and QSP — for precise clinical decisions
 기자가 쓴 기사 더보기
기자가 쓴 기사 더보기
A new company has emerged in Korea, combining AI and modeling technologies for the first time.
While numerous AI-driven drug development companies exist, few truly understand core clinical development areas like PK/PD and apply them in real-world drug development. APLUS Simulation, officially launched late last year, targets this gap.
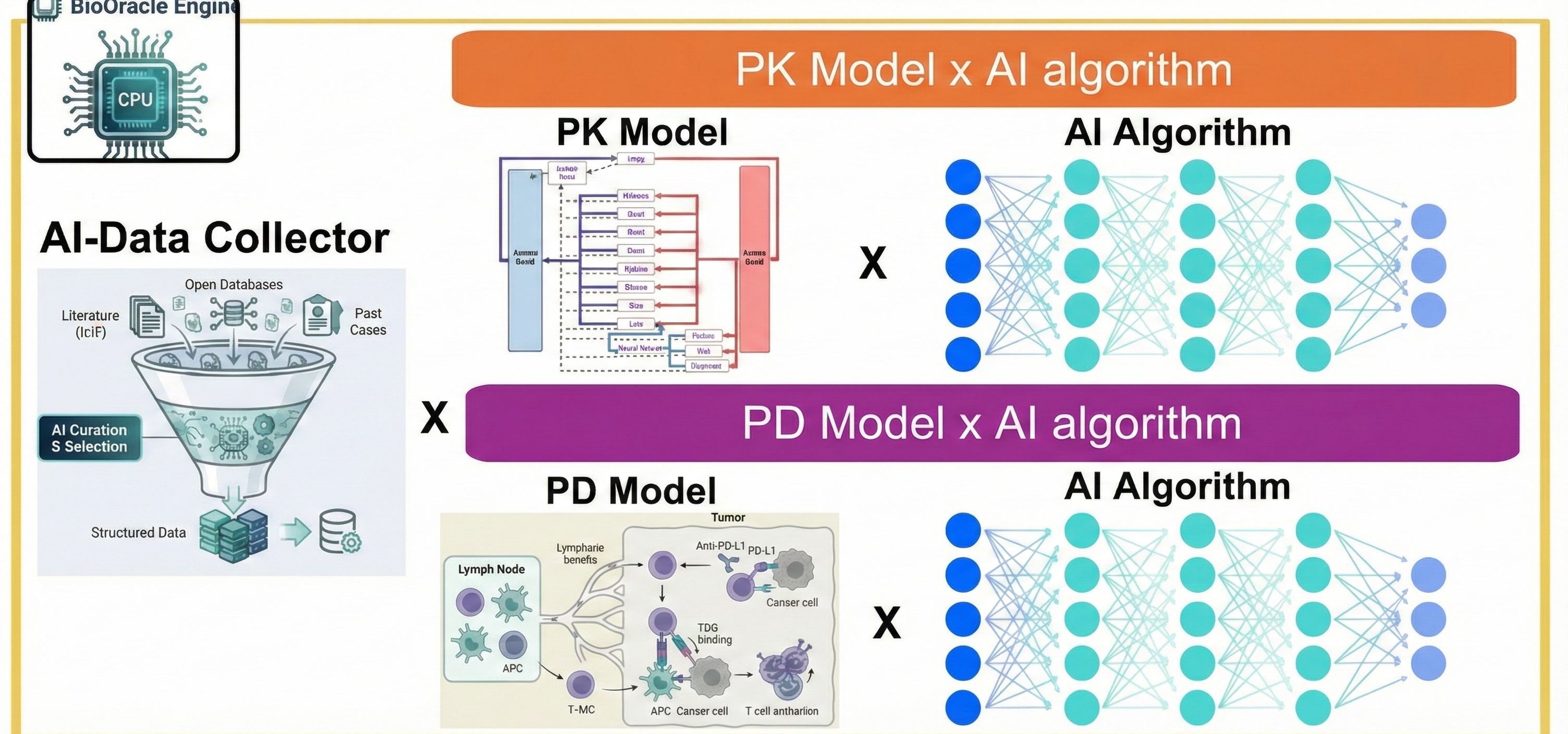
APLUS's core focus is integrating pharmacometrics-based modeling with AI technology. Building upon traditional pharmacometrics methodologies—PopPK, PBPK, and QSP centered on PK/PD—the company introduces new approaches by incorporating AI algorithms that add distinct value, ultimately enhancing clinical predictability and utility throughout drug development.
The company is developing computer-based AI-modeling software to support critical development decisions: AI algorithm-based automated data extraction and engine creation, animal-to-human translation prediction using AI and modeling, and ultimately, drug exposure and efficacy prediction across clinical trial phases.
Though newly launched, APLUS has deep industry roots. Co-founder and Co-CEO Soo Hyeon Bae (Ph.D.) and So Jin Lee (Pharm.D., Ph.D.) are both pharmacists who served as key consultants in pharmacometrics at Korea's first pharmacometrics modeling group. Dr. Bae gained experience across academia, public research institutions, and pharma industry, including an FDA fellowship program. Dr. Lee was a clinical pharmacist in the U.S. and a founding member of Korea's first modeling company and currently serves as adjunct professor at Sungkyunkwan University's School of Pharmacy. Together, they bring decades of combined expertise in pharmacometrics modeling and clinical pharmacology to APLUS.
APLUS's greatest strength lies in its AI-integration approach—using AI not as a black-box replacement for modeling, but as a tool to enhance mechanistic models that connect biological mechanisms and clinical data through equations. AI is selectively applied where clinical utility is needed: data extraction, identification of key factors affecting drug exposure and efficacy, model refinement, and prediction accuracy improvement.
Current projects focus on complex modalities including antibody drugs, bispecific antibodies, ADCs, and gene therapies, where understanding mechanisms of action is particularly critical and complex.
The company is already collaborating with multiple pharmaceutical and biotech companies as well as AI research teams. APLUS aims to provide practical value in drug development, deeply engaging with tailored solutions for drug development programs to ensure AI-modeling integration directly supports development decisions—ultimately increasing the efficiency and probability of drug development success.
This approach aligns with the regional government's recent emphasis on AI-driven drug development policy initiatives as well as the FDA's regulatory direction centered on NAMs (New Approach Methodologies), positioning APLUS at the forefront of the evolving landscape of AI and NAMs-based drug development.
| 인기기사 | 더보기 + |
-
1 에이비엘바이오 'ABL001' 쇼크?…“핵심은 그랩바디 플랫폼” -
2 메지온, FDA 공식 의견 제출...폰탄치료제 ‘JURVIGO’ 승인 가속 -
3 에이비엘바이오-컴퍼스, 담도암 2차 ‘토베시미그’ 2/3상 '무진행생존기간 개선' 확인 -
4 상장 제약·바이오 2025년 평균 자기자본이익률 코스피 0.30%·코스닥 -2.57% -
5 네이처셀,미국FDA 승인 알츠하이머병 임상 2b상 본격 개시 -
6 상장 제약·바이오 2025년 지배지분순이익 코스피 834억원·코스닥 72억원 -
7 “간의 벽 깨졌다” 올릭스 2.0, 비만·CNS siRNA 신약개발 드라이브 -
8 “조건부 허가 아니다” 큐로셀 CAR-T ‘림카토주’ 3상 없이 정식 허가된 이유 -
9 OLSS 서밋 2026, K-바이오 글로벌 도약의 산실로… 글로벌 창업 허브 향한 퀀텀 점프 -
10 경구 GLP-1 시장 개막…노보 ‘선점’, 릴리 ‘추격’
| 인터뷰 | 더보기 + |
| PEOPLE | 더보기 + |
| 컬쳐/클래시그널 | 더보기 + |

- Kwon, Hyeok Jin 기자 hjkwon@yakup.com
- 입력 2026.02.05 06:00 수정 2026.02.05 23:10

A new company has emerged in Korea, combining AI and modeling technologies for the first time.
While numerous AI-driven drug development companies exist, few truly understand core clinical development areas like PK/PD and apply them in real-world drug development. APLUS Simulation, officially launched late last year, targets this gap.
APLUS's core focus is integrating pharmacometrics-based modeling with AI technology. Building upon traditional pharmacometrics methodologies—PopPK, PBPK, and QSP centered on PK/PD—the company introduces new approaches by incorporating AI algorithms that add distinct value, ultimately enhancing clinical predictability and utility throughout drug development.
The company is developing computer-based AI-modeling software to support critical development decisions: AI algorithm-based automated data extraction and engine creation, animal-to-human translation prediction using AI and modeling, and ultimately, drug exposure and efficacy prediction across clinical trial phases.
Though newly launched, APLUS has deep industry roots. Co-founder and Co-CEO Soo Hyeon Bae (Ph.D.) and So Jin Lee (Pharm.D., Ph.D.) are both pharmacists who served as key consultants in pharmacometrics at Korea's first pharmacometrics modeling group. Dr. Bae gained experience across academia, public research institutions, and pharma industry, including an FDA fellowship program. Dr. Lee was a clinical pharmacist in the U.S. and a founding member of Korea's first modeling company and currently serves as adjunct professor at Sungkyunkwan University's School of Pharmacy. Together, they bring decades of combined expertise in pharmacometrics modeling and clinical pharmacology to APLUS.
APLUS's greatest strength lies in its AI-integration approach—using AI not as a black-box replacement for modeling, but as a tool to enhance mechanistic models that connect biological mechanisms and clinical data through equations. AI is selectively applied where clinical utility is needed: data extraction, identification of key factors affecting drug exposure and efficacy, model refinement, and prediction accuracy improvement.
Current projects focus on complex modalities including antibody drugs, bispecific antibodies, ADCs, and gene therapies, where understanding mechanisms of action is particularly critical and complex.
The company is already collaborating with multiple pharmaceutical and biotech companies as well as AI research teams. APLUS aims to provide practical value in drug development, deeply engaging with tailored solutions for drug development programs to ensure AI-modeling integration directly supports development decisions—ultimately increasing the efficiency and probability of drug development success.
This approach aligns with the regional government's recent emphasis on AI-driven drug development policy initiatives as well as the FDA's regulatory direction centered on NAMs (New Approach Methodologies), positioning APLUS at the forefront of the evolving landscape of AI and NAMs-based drug development.
무단 전재·복사·배포 등을 금지합니다.



